Abstract:
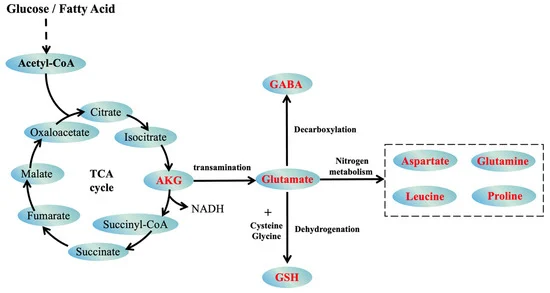
This groundbreaking article presents a unified theory of aging that integrates evolutionary biology, epigenetics, and metabolic regulation, offering a paradigm shift in our understanding of senescence. By synthesizing recent research on Horvath’s epigenetic clock, the GABA-glutamate-αKG axis, and the evolutionary layers of aging systems, the authors propose a compelling model where hormonal changes trigger the expression of short LARP1, a key orchestrator of four distinct aging systems. The theory elucidates how luteinizing hormone (LH), human chorionic gonadotropin (hCG), and follicle-stimulating hormone (FSH) differentially activate these systems, explaining gender-specific aging patterns. Furthermore, it reveals the unexpected role of SP-1 in linking sexual maturation to aging through regulation of MAO-A, MAO-B, and WRN. This comprehensive framework not only explains the acceleration of aging but also identifies novel therapeutic targets, potentially revolutionizing anti-aging interventions. By connecting hormonal changes, metabolic imbalances, and epigenetic dysregulation into a cohesive aging program, this article challenges long-held beliefs about the nature of aging and opens new avenues for extending healthspan and lifespan1.