Key Points
Research suggests Bowles’s APES theory, focusing on Aging, Predation, Extinction, and Sex, may outperform the modern evolutionary synthesis in explaining aging and reproductive strategies.
It seems likely that the APES theory better accounts for programmed aging, lifespan variations based on predation defense, and male sex traits as predator attractants, challenging the modern synthesis’s dominance.
The evidence leans toward the APES theory’s son-king hypothesis for menopause, supported by historical figures like Ramses (93 children) and Genghis Khan (A large percentage of Asian males share his genes), contrasting with the grandmother hypothesis, which Bowles argues is disproven.
An unexpected detail is that the APES theory explains asexual animals in low-predation environments and homosexuality linked to prenatal stress, with studies on rats, mice, and WW2 Germany supporting this.
Abstract:
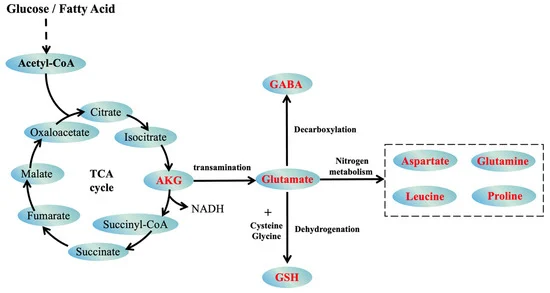
This groundbreaking article presents a unified theory of aging that integrates evolutionary biology, epigenetics, and metabolic regulation, offering a paradigm shift in our understanding of senescence. By synthesizing recent research on Horvath’s epigenetic clock, the GABA-glutamate-αKG axis, and the evolutionary layers of aging systems, the authors propose a compelling model where hormonal changes trigger the expression of short LARP1, a key orchestrator of four distinct aging systems. The theory elucidates how luteinizing hormone (LH), human chorionic gonadotropin (hCG), and follicle-stimulating hormone (FSH) differentially activate these systems, explaining gender-specific aging patterns. Furthermore, it reveals the unexpected role of SP-1 in linking sexual maturation to aging through regulation of MAO-A, MAO-B, and WRN. This comprehensive framework not only explains the acceleration of aging but also identifies novel therapeutic targets, potentially revolutionizing anti-aging interventions. By connecting hormonal changes, metabolic imbalances, and epigenetic dysregulation into a cohesive aging program, this article challenges long-held beliefs about the nature of aging and opens new avenues for extending healthspan and lifespan1.